Ivydene Gardens Soil: |
||
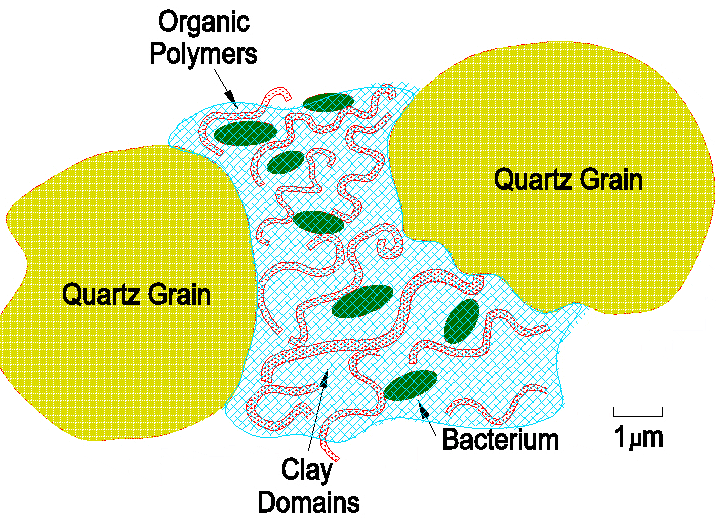
Soil Structure This describes the way in which sand, silt and clay particles are bonded together in larger units called ‘aggregates’. These are formed when the soil is subjected to shrinking and swelling, plant-root penetration or freezing. All these processes tend to break the soil into discrete units. Aggregates are said to be stable when they are able to resist pressures caused by processes such as compaction and sudden wetting. Rapid wetting is a process in breaking up unstable aggregates, because when dry aggregates are suddenly exposed to water, pores near the surface of the aggregate become filled with water, trapping air inside the aggregate; the resulting pressure can sometimes be enough to break the aggregate apart, and this is called ‘slaking’. Aggregates are divided into microaggregates (less than 250 millionths of a metre) and macroaggregates (greater than 250 millionths of a metre). Before microaggregates can form, microscopic clay minerals need to be grouped together in small stacks called ‘domains’. When clays are bonded together in this way, they are termed ‘flocculated’. The most important factor influencing flocculation is the presence of ions with more than 1 charge. When clay minerals are covered with singly charged ions they disperse and become deflocculated (i.e. they will absorb a great deal of water without it draining).
However, not all ions carry only 1 charge. For example, calcium (Ca2+) in lime or chalk, Magnesium (Mg2+) and aluminium (Al3+) are 3 very common ions in soils. Ions with multiple charges allow clay minerals to bond together to form domains. Once clay minerals are stacked together to form domains, they can then bond with organic matter to form microaggregates. |
||
The interaction between clay domains, organic matter, silt and sand particles diagram.
|
||
|
Site design and content copyright ©December 2006. Page structure amended September 2012. Menu tables amanded July 2015 by Chris Garnons-Williams. DISCLAIMER: Links to external sites are provided as a courtesy to visitors. Ivydene Horticultural Services are not responsible for the content and/or quality of external web sites linked from this site. |
Once microaggregates have formed, they can then coalesce to form macroaggregates. In soils that have low concentrations of clay, macroaggregate stability is highly dependent on organic matter. The type of organic matter associated with macroaggregates is slightly different from the persistent organic material found in microaggregates. Type one are those stabilising agents that are referred to as ‘temporary’. These consist of microbial and plant by-products, the most important of which are the ‘polysaccharide gums’ that are simply long chains of sugar molecules. Secondly, there are ‘transient’ stabilising agents, which include the fine plant roots and fungal hyphae. Both stabilising compounds are vulnerable to microbial attack so need to be replenished continuously through inputs of fresh soil organic matter.
If microaggregates do not have a continuing supply of organic matter, then they will break up so that soil particles simply return to being sand, silt or clay. |
|
Bonding Family and Friends "...even those for whom cooking is an oppressive chore or a source of self-doubting anxiety, achnowledge that a meal shared by friends and family is one of the bonding rituals without which the family, society even, can fall apart." by Antonia Till from "Loaves & Wishes". |
||
The following information comes from pages 30-34 of the 844 in Soils may be said to consist of a mechanical mixture of 4 substances -
with many of the following 11 chemical substances , in varying proportions:- |
||
Chemical |
is contained in or made from |
Benefit |
Potash - is the common term for nutrient forms of the element potassium (K) |
This substance is obtained by burning wood, small branches, or leaves, the ash being washed in water, and evaporated in an iron pot and calcined. Add a small quantity of water, decant the liquid, and evaporate to dryness, and pearl-ash is obtained, which is an impure form of potash in combination with carbonic acid, or crude carbonate of potash. When this is boiled with newly-slaked quicklime, it is deprived of carbonic acid, which enters into combination with the lime, and the carbonate of potash is thus converted into pure or caustic potash, which can be separated into a silvery-white soft, metallic substance, potassium, and a gaseous element, oxygen. The combination in which potash is found in soils is chiefly as silicates of potash. Some kinds of felspar, mica, and granite contain large proportions, as much as 15-20%. Iit also enters into the composition of trap-rock, basalt, and whinstone, though in smaller proportions. |
Many plants require a large amount of potash for their food, the only source from which it can be obtained being the soil. This accounts for the fact that wood ashes, which contain carbonate of potash, are so conducive to the healthy growth of clover, beans, potatoes, and other plants whose ashes yield potash in return. As the rock crumbles, silicates of potash are set free, and rendered available for the plants. Clay, which is chiefly derived from felspar, invariably contains it; and it is partly for this reason that light land, in which potash is usually deficient, is benefitted by claying. |
This is obtained by burning seaweed; and plants growing on the sea-shore are rendered caustic by the same process. |
Its most common form, however, is sea-salt, or chloride of sodium. Seakale, asparagus, and similiar plants are benefitted by its use. |
|
Lime - is a calcium-containing (Ca2+) inorganic material in which carbonates, oxides and hydroxides predominate. |
Chalk, marble, and limestone are carbonates of lime. Under heat, the carbonic acid is driven out, and pure or caustic lime (Calcium oxide, CaO) remains. Quicklime sprinkled with water absorbs it; heat is evolved, and it falls to powder, or is slaked. Slaked lime is a white powder, dry to appearance, but contains, in reality, water in an invisible form, chemically combined with lime. If exposed to the air, it attracts carbonic acid from the atmosphere, and becomes partially changed into carbonate of lime. |
In its effects on animal and vegetable matters this pure or caustic lime resembles potash and soda, is slower in action, and is used most beneficially on peat land; its excess of organic matter is thus gradually destroyed, and converted into nutritious food for plants. Salts of lime are found in all ashes of plants; soils, therefore, capable of sustaining vegetable life, must contain lime in some form or other. |
Magnesia - a natural mineral of magnesium oxide (Magnesium Mg2+) |
The ingredient is never wanting in fertile soils. Magnesian limestone, which is a natural compound of the carbonates of lime and magnesia, contains 30-40%; and in this form it exists in all dolomite and many other solid rocks. Soils containing much carbonate of magnesia absorb moisture with great avidity, and are generally cold soils. Silicate of magnesia enters largely into the composition of serpentine rocks. Soapstone and limestone frequently contain it. Compounds of sulphuric acid and muriatic acid with magnesia are also found in many mineral waters. |
Suphate of magnesia, which is the name of the familiar Epsom salts, is formed from the decomposition of dolomitic rocks. |
Alumina - Aluminium oxide (Al2O3). |
This is the compound of the metal aluminium with oxygen, or, in other words, oxide of aluminium. It occurs very abundantly in the mineral kingdom, both free and in combination with acids. In its crystallized state it forms the hard mineral known as corundum, and, in combination with oxide of chromium, the saphire and the ruby; and emery is a dark-coloured granular variety of it. In an uncrystallized state it is a white, tasteless, powdery substance, obtained by adding a solution of carbonate of soda to alum. |
It constitutes a large proportion of shale and slate rocks, and is a principle ingredient, in combination with silica, in pipe, porcelain, and agricultural clays, to which it gives tenacity and stiffness. It is rarely found in the ashes of plants, and therefore not considered as directly contributing to their nourishment, although useful as a mechanical agent in absorbing ammonia from the atmosphere, and in detaining the volatile as well as the alkaline salts of manures, which would otherwise be dissolved by the first heavy shower, and carried into the subsoil beyond the reach of the roots of the plant. |
Iron - is a chemical element with symbol Fe (from Latin: ferrum). Iron compounds are called ferrous. |
This metal, both in the black or protoxide, and the red or peroxide state, abounds in all soils, the red being most abundant, and easily observable from the red colour it communicates. Even soils in which the protoxide obtains, which are a bluish-grey colour when brought to the surface, are changed to the red colour by the atmosphere, oxygen uniting with and acting on it. Oxide of iron is found in the ashes of all plants and in the blood of animals. |
The presence of iron is easily detected in soils by the ochry deposits in the beds of springs and ditches, where the oxide dissolved in carbonic acid produces the metallic-coloured deposit in question. Sulphate of iron also occurs in some soils, produced from iron pyrites: such soils are unproductive; for it is a compound of sulphuric acid with protoxide of iron, better known under the name of green vitriol. Lime added to such soils combines with the sulphuric acid, forming gypsum; and sweetens them and removes the injurious properties. |
Manganese - Manganese is a chemical element with symbol Mn. It is not found as a free element in nature; it is often found in combination with iron, and in many minerals. Manganese is also important in photosynthetic oxygen evolution in chloroplasts in plants. The oxygen-evolving complex (OEC) is a part of photosystem II contained in the thylakoid membranes of chloroplasts; it is responsible for the terminal photooxidation of water during the light reactions of photosynthesis, and has a metalloenzyme core containing four atoms of manganese. For this reason, most broad-spectrum plant fertilizers contain manganese. |
This metal, in combination with oxygen, associated with oxide of iron, occurs naturally in many soils. |
In the ashes of plants traces of it are also found; but iron usually predominates. The ash of the horse chestnut and oak bark is rich in manganese, with no trace of iron. |
Silica or Silex - Silicon dioxide, also known as silica (from the Latin silex), is a chemical compound that is an oxide of silicon with the chemical formula SiO2. |
This mineral occurs abundantly in nature, either in a free state or in the form of sand, sandstones, flint, chalcedony, rock-crystal, or quartz, and in combination with lime, magnesia, iron, potash, soda, and other minerals. Silica is
|
Silica is dissolved, or rather enters into combination with the alkali, and forms glass; or when the alkali is in excess, it dissolves into water. On the addition of muriatic acid, or sulphuric acid, to a solution of this silicate of potash, the silcate separates into a gelatinous mass, in which form it is soluble in water, and thus becomes the food of plants. |
Sulphur - sulphur (sulfur) is a chemical element with symbol S. Sulphur is an essential element for all life, but almost always in the form of organosulfur compounds or metal sulfides. Sulfur is increasingly used as a component of fertilizers. The most important form of sulfur for fertilizer is the mineral calcium sulfate. Elemental sulfur is hydrophobic (that is, it is not soluble in water) and, therefore, cannot be directly utilized by plants. Over time, soil bacteria can convert it to soluble derivatives, which can then be utilized by plants. Sulfur improves the use efficiency of other essential plant nutrients, particularly nitrogen and phosphorus. Biologically produced sulfur particles are naturally hydrophilic due to a biopolymer coating. This sulfur is, therefore, easier to disperse over the land (via spraying as a diluted slurry), and results in a faster release. Plant requirements for sulfur are equal to or exceed those for phosphorus. It is one of the major nutrients essential for plant growth, root nodule formation of legumes and plants protection mechanisms. Sulfur deficiency has become widespread in many countries in Europe. |
This compound, in the form of sulphuric acid, enters into the composition of all cultivated soils, chiefly in combination with limestone, magnesia, potash, and other bases. |
With hydrogen it forms sulhuretted hydrogen, a remarably disagreeable-smelling gas, the product of the decompostion of organic matter contained in the soil and impregnating many medicinal waters, as at Harrogate in North England. |
Phosphorus - Phosphorus is a chemical element with symbol P . Phosphorus is essential for life. Phosphates (compounds containing the phosphate ion, PO4−3) are a component of DNA, RNA, ATP, and also the phospholipids, which form all cell membranes. Demonstrating the link between phosphorus and life, elemental phosphorus was first isolated from human urine, and bone ash was an important early phosphate source. Phosphate minerals are fossils. Low phosphate levels are an important limit to growth in some aquatic systems. Phosphate is needed to replace the phosphorus that plants remove from the soil, and its annual demand is rising nearly twice as fast as the growth of the human population. |
This ingredient is a soft, wax-like, highly inflammable substance, which combines with atmospheric oxygen, giving rise to phosphoric acid, which enters into the composition of all our cultivated plants, and is essentially necessary to a healthy condition of vegetable life. |
Phosphorus exists in trap-rock, granite, basalt and other igneous rocks, and in lime, ironstone and most minerals. |
Chlorine - Chlorine is a chemical element with symbol Cl. In the form of chloride ions, chlorine is necessary to all known species of life. Chloride is one of the most common anions in nature. |
This is a highly-noxious, suffocating, yellowish, gaseous element, particularly disagreeable in smell. |
In soils it is found in combination with such bases as chloride of sodium, or common salt. It is more necessary as a plant-food to root crops rather than to cereals. |
|
Some of the affects of the above 11 chemicals on the following soils:- |
|
|
|
|
Sandy Soils - If you try to adjust the soil texture by adding silt or clay to a sandy soil, you’ll see some improvement, but most of it will just flush through the soil. There’s not enough organic matter to keep these fine-textured soil components from washing out. Increasing soil organic matter is the key to gardening in sandy soil. You have to make the soil more “sticky”, so water and nutrients don’t just flush through every time it rains. My suggestion:- |
Sandy soils are loose, friable, open and dry, and for that reason easily cultivated. They rest chiefly on the old red sandstone, and granite and coal formations. |
Where alumina and calcareous matter are absent, however, they are nearly barren, they absorb manures without benefit to the land. Where alumina and lime exist, they are more compact and adhesive, and grow good crops of beans, peas, spring wheat, and turnips. They are capable of improvement by admixture with clay, marl, chalk, and other adhesive soils, which communicate their constituent properties to them |
Calcareous Soils - Calcareous soils have often more than 15% CaCO3 (Calcium carbonate) in the soil. Phosphorous is often lacking in calcareous soils. Calcareous soils usually suffer from a lack of micronutrients, especially zinc and iron. Heavy applications of animal manure are helpful in preventing deficiency of iron and zinc. Calcareous is an adjective meaning "mostly or partly composed of calcium carbonate", in other words, containing lime or being chalky. Calcareous soils are relatively alkaline, in other words they have a high pH. The availability of N, P, K, Mg, Mn, Zn, and Fe to citrus decreases when soil CaCO3 concentration increases to more than about 3% by weight. These soils generally have a pH value in the range of 7.6 to 8.3. Phosphorus fertilizer applied to calcareous soils becomes fixed in sparingly soluble compounds over time. To maintain continuous P availability, P fertilizer should be applied on a regular, but not necessarily frequent, basis. |
Calcareous soils resting on the upper chalk formation are usually deep, dry, loose, friable, and fertile in their nature, but others, resting on the shaly oolite, are stony, poor, thin soils. Where pure clay is present in such soils, they are called loams or calcareous clays; where silica is in excess, they are termed calcareous sandy soils. |
Leguminous plants, as peas, beans, vetches, saintfoin, and clover, do well on such soils, lime being essential to their growth. |
Clay Soils - Interesting 4 page article applying compost etc to improve clay soil. To improve that soil as the quickest solution, I would add a 1 cm (0.5 inch) depth of sharp washed sand, a 1 cm depth of chalk (lime) and 10 cm (4 inch) depth of organic compost and leave it to work itself in from that application in early spring. 2 months later add 3 inches (7.5 cm) depth of cow manure to provide further nutrients. |
Clay soils are characterised by stiffness, impenetrability, great power of absorbing and retaining moisture, and great specific gravity; they are, consequently, cold, stiff, heavy, and impervious, costly to cultivate, and often unproductive. |
Perfect drainage, |
|
The following weeds indicate what the soil is that they naturally grow on |
|
Argillaceous or Clayey Soil |
Common coltsfoot indicates blue clay
|
Tussilago farfara The most certain and universal sign of a clayey soil, and the chief plant found on alum grounds of Britain, France, and Italy |
Goose tansy (Goosegrass) |
Potentilla anserina |
|
Silvery-leaved tansy (Hoary cinquefoil) |
Potentilla argentea |
|
Creeping tansy (Creeping cinquefoil) |
Potentilla reptans |
|
Yellow meadowrue (Common meadow-rue) |
Thalictrum flavuum |
|
Carex many species |
||
Juncus various species |
||
Tuberous bitter vetch (Bitter vetch, Heath Pea) |
Orobus tuberosus (Lathyrus linifolius subsp. montanus) |
|
Greater bird's-foot trefoil
|
Lotus major (Lotus uliginosus) |
|
Small-horned trefoil (Common birdsfoot trefoil) |
Lotus corniculatus |
|
Common soapwort (Soapwort)
|
Saponaria officinalis |
|
|
|
|
Calcareous Soil |
Spiked speedwell |
Veronica spicata |
Little bedstraw |
Galium pusillum |
|
Common gromwell
|
Lithospermum officinal |
|
Purple-blue gromwell (Purple gromwell)
|
Lithospermum purpureo-caeruleum |
|
Clustered bell-flower
|
Campanula glomerata |
|
Hybrid bell-flower (Venus's Looking-glass) |
Specularia hybrida (Legousia hybrida) |
|
Round-headed rampion
|
Phyteuma orbiculare (Phyteuma tenerum) |
|
Lychnitis mullein (White Mullein) |
Verbascum lychnitis |
|
Wayfaring tree
|
Viburnum lantana |
|
Common berberry (Barberry) |
Berberis vulgaris |
|
Common dwarf sun-rose (Common Rock-Rose) |
Helianthemum vulgare (Helianthemum nummularium, Helianthemum chamaecistus) |
|
Common pulsatilla anemone (Pasque Flower)
|
Anemone pulsatilla |
|
White vine, virgin's bower or traveller's joy
|
Clematis vitalba |
|
Cultivated sainfoin (Sainfoin) |
Onobrychis sativa (Onobrychis viciifolia) |
|
|
|
|
Sandy or Siliceous Soil |
Veronica triphylla (Veronica triphyllos) |
|
Spring speedwell |
Veronica verna |
|
Italian viper's bugloss (Pale Bugloss) Echium plantagineum contains pyrrolizidine alkaloids and is poisonous.[10] When eaten in large quantities, it causes reduced livestock weight and death, in severe cases. Paterson's curse can kill horses[11] and irritate the udders of dairy cows and the skin of humans. After the 2003 Canberra bushfires a large bloom of the plant occurred on the burned land, and many horses became ill and died from grazing on it.[12] Because the alkaloids can also be found in the nectar of Paterson's curse, the honey made from it should be blended with other honeys to dilute the toxins. |
Echium italicum |
|
Smooth rupture wort |
Herniaria glabra |
|
Hairy rupture wort - Herniaria hirsuta is a species of flowering plant in the pink family known by the common name hairy rupturewort. It is native to Eurasia and North Africa, and it is known on other continents, including North America, as an introduced species. |
Herniaria hirsuta |
|
English catchfly (Small-flowered Catchfly) |
Silene anglica and other species |
|
Red sandwort indicates poor sand |
Arenaria rubra |
|
Cornfield spurrey (Corn spurrey)
|
Spergula arvensis |
|
Hybrid poppy (Bristly Poppy) |
Papaver hybridum |
|
Scarlet poppy (Pale Poppy) |
Papaver argemone |
|
|
|
|
Ferruginous Soil - Ferrallitisation is the process in which rock is changed into a soil consisting of clay (kaolinite) and sesquioxides, in the form of hydrated oxides of iron and aluminium. In humid tropical areas, with consistently high temperatures and rainfall for all or most of the year, chemical weathering rapidly breaks down the rock. This at first produces clays which later also break down to form silica. The silica is removed by leaching and the sesquioxides of iron and aluminium remain, giving the characteristic red colour of many tropical soils. Ferrallitisation is the reverse of podsolisation, where silica remains and the iron and aluminum are removed. In tropical rain forests with rain throughout the year, ferrallitic soils develop. In savanna areas, with altering dry and wet climates, ferruginous soils occur. |
Garden sorrel indicates the presence of iron or peat (Sheep's Sorrel) |
Rumex acetosa |
|
|
Oxalis acetosella |
|
|
|
|
Peaty Soil - This kind of soil is basically formed by the accumulation of dead and decayed organic matter, it naturally contains much more organic matter than most of the soils. It is generally found in marshy areas. Now the decomposition of the organic matter in Peaty soil is blocked by the acidity of the soil. This kind of soil is formed in wet climate. Though the soil is rich in organic matter, nutrients present are fewer in this soil type than any other type. Peaty soil is prone to water logging but if the soil is fertilized well and the drainage of the soil is looked after, it can be the ideal for growing plants. |
|
Vaccinum myrtillus |
Bleaberry (Northern bilberry) |
Vaccinum uliginosum |
|
Oxycoccus palustris |
||
Heath - See Heath Family and Erica |
Erica |
|
Awl leaved spurrey (Heath Pearlwort) |
||
Officinal septfoil (Tomentil) |
Tormentilla officinalis (Potentilla erecta) |
|
|
|
|
Saline or Salt Soil - Researchers at the University of California at Davis are breeding barley for culture with sea water irrigation (Epstein, 1976). Lines have been developed which survive and set seed (yields in the order of 1188 kg/ha) under irrigation with undiluted sea water. Similar breeding is underway with wheat. Table 18 states tolerance of fruit varieties and root stocks to Chloride levels. Leaching is the only practical way of removing excess salts. This is effective only to the extent that water moves down through the soil profile and beneath the root zone (drainage must be good). The amount of salts removed depends on the quantity and quality of water leached through the soil profile during a single irrigation period. Water should be low in salts (high quality) and must not run off the surface. It should be applied slowly so amounts do not exceed the ability of the soil to take in water (infiltration rate). The following amounts of water applied in a single, continuous irrigation will dissolve and decrease soil salts by these fractional amounts: • 6 inches of water will leach about ½ the salt • 12 inches of water will leach about 4/5 of the salt. • 24 inches of water will leach about 9/10 of the salt. Salty soils are not reclaimable when the soil’s clay content, compaction, or hardpan prevents leaching. |
Glasswort - Glassworts Family
|
Salicornia |
Marine wrackgrass (Common eel-grass) |
Zosteria marina |
|
Sea ruppia (Tassel Pondweed) |
Ruppia maritima |
|
Sea lungwort (Oyster Plant) |
Pulmonaria maritima (Mertensia maritima) |
|
Soldanella-leaved beanbind (Sea Bindweed) |
Calystegia soldanella |
|
Whorled knotgrass (Coral necklace) |
Illecebrum verticillatum |
|
Sea goosefoot (Common seablight) |
Chenopodium maritimum (Suaeda maritima) |
|
Shrubby goosefoot (Shrubby seablite) |
Chenopodium fruticosum (Suaeda fruticosa) |
|
Kali saltwort (Saltwort) |
Salsola kali |
|
Sison verticillatum |
||
Marine sandwort |
Arenaria marina |
|
Fringed orach (Frosted orache) |
Atriplex laciniata |
|
|
|
|
Aquatic or Moist Soil - Standing water under wading bird rookeries is critical to limiting predation and enhancing nest success. Draining impoundments while wading birds are actively nesting is strongly discouraged, regardless of other management needs. Appendix 1 gives a waterfowl food value guide for common moist-soil plants in the Southeast of USA. |
|
Caltha palustris |
Common mare's-tail (Marestail) |
Hippuris vulgaris |
|
|
|
Pinguicula vulgaris |
|
European water-horehound (Gipsywort) |
Lycopus Europeus |
|
Dioecious valerian (Marsh valerian) |
Valeriana dioica |
|
|
|
Hottonia palustris |
|
Valerandi's brookweed (Brookweed)
|
Samolus valerandi |
|
Marsh thysselinum (Milk parsley) |
Thysselium palustre (Peucedanum palustre) |
|
Square-stalked willow herb |
Epilobium tetragonum |
|
Willow-like lythrum (Purple Loosestrife) |
Lythrum salicaria |
|
Tongue-leaved crowfoot (Greater Spearwort) |
Ranunculus lingua |
|
Spearwort |
Ranunculus flammeus |
|
|
|
|
Very Dry Soil - Drought-resistant plants. Overcoming dry soils in Australia with list of drought-tolerant plants. |
Red sandwort indicates poor sand |
Arenaria rubra |
Garden sorrel (Common sorrel) |
Rumex acetosa |
|
|
|
Thymus serpyllum |
|
Basil-leaved or common acynos, a thyme-like plant (Basil thyme) |
Acynos vulgaris (Acynos is Basil-thyme and Acynos vulgaris is Common basil-thyme. |
|
Field trefoil (Haresfoot clover) |
Trifolium arvense |
|
|
|
|
|
|
|
|
||
SOIL PAGE MENU Soil Introduction - How does Water act in Soil SOIL SUBSIDENCE
|
Click on Black or White box in Colour of Month. |
LATE SUMMER GALLERY PAGES FOLIAGE COLOUR BULB, CORM, RHIZOME AND TUBER INDEX - There are over 700 bulbs in the bulb galleries. The respective flower thumbnail, months of flowering, height and width, foliage thumbnail, |
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Besides the above Bulb Flower Colour Comparison Pages, you also have the following Comparison Pages:- |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Late Summer INDEX link to Bulb Description Page |
Flower Colour with Flower Thumbnail |
Flowering Months Mat, |
Height x Width in inches (cms) - Seed Head Thumbnail Soil Sun Aspect Soil Moisture |
Foliage Colour |
Bulb Use |
Comments |
PLANTS PAGE PLANT USE Groundcover Height Poisonous Cultivated and UK Wildflower Plants with Photos
Following parts of Level 2a, |
PLANTS PAGE MENU
|
PLANTS PAGE MENU
Photos - 12 Flower Colours per Month in its Bloom Colour Wheel Gallery
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Acis "They are excellent for cutting and make a good display either in a bed or in a thin woodland. They also do quite well in grass, which must not be mown until their leaves begin to die down. "Indoor Culture in Window-boxes - Plant in clumps during October, 3 inches (7.5 cms) deep, 2 inches (5 cms) apart. These are excellent for a site in partial shade, but will only succeed if left undisturbed for 2 or 3 years. Suitable varieties are Leucojum aestivum 'Gravetye Giant' and Leucojum vernum." from Indoor Bulb Growing by Edward Pearson. Published by Latimer House Limited in 1953. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Acis autumnalis |
White
|
August, September, |
4-6 x 4 |
Dark Green grass-like foliage, often being produced shortly after the flower spike. |
Plant at edge of bed. Use in rock garden. Cut flower. Thin woodland or shade from shrubs. Naturalize in grass. |
In autumn it throws up leafless stems from which it bears 2-4 bell shaped white flowers, often with red bases to them. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Acis autumnalis pulchellum - |
White
|
August, September, |
8 x 4 |
Dark Green grass-like foliage being produced at the same time as the flower spike. |
Plant at edge of bed. Use in rock garden. Cut flower. Thin woodland or shade from shrubs. Naturalize in grass. |
Plant with 1 or 2 inches (2.5 or 5 cms) of soil over the tops of the bulbs towards the front of a bed in an area where they can be left undisturbed. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Acis |
Pure White flowers on 4-8 inch stems
|
September, 6 petal, bell-shaped flowers in spike. |
4 x 2 |
Dark Green grass-like foliage being produced at the same time as the flower spike. |
Plant at edge of bed. Use in rock garden. Cut flower. Thin woodland or shade from shrubs. Naturalize in grass. |
Plant with 1 or 2 inches (2.5 or 5 cms) of soil over the tops of the bulbs towards the front of a bed in an area where they can be left undisturbed. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Acis valentinum |
White
|
February, March, |
10 x 12 |
Thin Grey-Green leaves being produced after the flower spike. |
Plant at edge of bed. Use in rock garden. Cut flow-er. Thin woodland or shade from shrubs. Naturalize in grass. Coastal conditions |
Grows in open, calcareous, stony and rocky places, hill slopes. Requires winter mulch to protect it from the worst of the weather. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
White with Red stripes
|
September, October, Umbel |
6-12 x 12 (15-30 x 30) Sand, Chalk |
Green cylindrical and hollow leaves |
These unusual autumn flowering species are ideal on a scree or rockery in full sun. They are hardy and also make nice pot specimens in a cold greenhouse. |
Native of the Pelo-ponnese. Plant at soil level and 4 inches (10 cms) apart. All Alliums have the distinctive onion smell, both in the foliage and bulb. This smell can be used to reduce aphid infestations on flowers by planting 1 each side of the infected plant. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Babiana stricta - tender |
Pale Cream through Purple, Mauve and Blue and Crimson
|
March, April, May 5 petal, funnel-shaped flowers in a spike with slight fragrance |
6-18 x 4 |
Sword-shaped 5 inches (12.5 cm) long, 0.5 inches (1.125 cms) wide, green
|
Plant against South-facing House Wall in Southern England where temperatures do not go below -5 degrees Centigrade. Mulch with 3 inches (7.5 cms) of organic compost to conserve moisture in the summer. |
Set 6 inches (15 cms) deep in average and sandy soils, a little shallower in heavy clay - put 2 inches (5 cms) of sand surrounding bulb to prevent rotting - soils, 6 inches (15 cms) apart. Leave undisturbed for years. Remove mulch during autumn and winter. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Dark Green to Dark Brown Spathe
|
September, October, November Up to 6 inches (15 cm) long spathe but not a flower |
4-8 x 12 Scree, Sand or Chalky soil with 1 inch (2.5 cms) of sand worked into the top 2 inches (5 cms). |
The 5-10 light green leaves are 1 inch wide and 2-4 inches long.
|
Can be planted beside a path in a rock garden where it is is a rocky, sandy location in full sun in Southern England. |
Biarum is a group of unusual looking bulbs, grown for their weird and wonderful spathes that are produced in autumn. Not fully hardy so these are best grown in pots in the garden before spending the winter in a greenhouse. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Light Green with
|
September, October Up to 6 inches (15 cm) long spathe but not a flower |
3-4 x 12 |
5-10 light Green leaves emerge in Sep-Oct
|
Can be planted beside a path in a rock garden where it is is a rocky, sandy location in full sun in Southern England. |
Not fully hardy so these are best grown in pots in the garden before spending the winter in a greenhouse. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Biarum tenuifolium |
Pale Green with Purple Flush Spathe
|
July, August, September, October, November |
10 x 12 |
5-10 light Green leaves emerge in Sep-Oct
|
Can be planted beside a path in a rock garden where it is is a rocky, sandy location in full sun in Southern England. |
Native to the central and eastern Mediterranean. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Bright Green with
|
September Up to 6 inches (15 cm) long spathe but not a flower |
9 x 12 |
5-10 light Green leaves emerge in Sep-Oct |
Can be planted beside a path in a rock garden where it is is a rocky, sandy location in full sun in Southern England. |
Native to Northern Greece and Italy. Not fully hardy so these are best grown in pots in the garden before spending the winter in a greenhouse. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
"The Erythroniums native to the Western U.S. are considered by many to be the most beautiful of the genus. Often called "Fawn Lilies" because of the dappled coloring to the leaves, they have dainty nodding flowers like small lilies, set off by large shining leaves that are either plain green or marbled with silver and bronze. Most grow in shaded woodland areas that go quite dry in summer, but with excellent drainage, they can tolerate some summer water." from Telos Rare Bulbs in USA. "Culture in Garden - They like a damp, well-drained soil, and a partially drained position. The bulbs must not be kept out of the ground any longer than necessary, as they resent being moved, nor must the best results be expected at their first time of flowering. It follows that they should be left alone as long as they flower well. An anual top-dressing of a mixture of light decayed manure and peat benefits them. They are increased by offsets and by seed, which last should be thinly sown in pans in a cold frame in August, and the seedlings grown on for 2 years before planted out in the the open; or if room can be found, in loose soil in a cold frame where they remain until the bloom, when the best can be marked before they are put in their permanent places." from Black's Gardening Dictionary. Edited by E.T. Ellis, F.R.H.S. Second edition. Published by A. & C. Black Ltd. in 1928. "The largest flower spikes are found where the ground has recently been burnt, so it is possible that a top dressing of potash would have the same effect. If they are to be divided and moved in the same garden this is probably best done when they are beginning to die down after flowering." from Collins Guide to Bulbs by Patrick M. Synge. Reprinted 173. ISBN 0 00 214016-0 "Suitable for cultivation in the garden, greenhouse or house. They succeed in any good well-drained garden soil, but the ideal compost is equal parts loam, peat, leaf mould and sand. The bulbs should be planted in August in a shady position in beds, rock gardens, edges or under trees. Once planted, they need not be disturbed for many years. "Rock Garden Culture for Erythronium citrinum (Yellow flowers); Erythronium Frans Hals (Purple-rose flowers); Erythronium revolutum (Pink flowers); Erythronium Hartwegii (Creamy-white flowers) - Plant in September 1.5 inches (3.75 cms) deep and 4 inches (10 cms) apart, in partial shade, in moist, well-drained sandy loam and ample leaf-mould or peat. Surround the tubers with about an inch (2.5 cms) of silver sand, and do not lift more often than necessary, but mulch annually with well-rotted manure and leaf-mould. Propagate by means of seed in a frame in August. Thin out but do not plant the seedlings out until the third September after sowing. The plants are also increased by offsets." from Rock Gardens how to plan and plant them with sections on the Wall, Paved, Marsh and Water Gardens by A. Edwards in charge of the rock garden, kew. Published by Ward, Lock & Co. in 1929. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Erythronium |
White,
Each flower stem will have 1-10 downward pointing flowers, with reflexed petals. |
April, May, June Clump. |
6 x 5 Humus-rich Sand. Bulbs must be kept slightly damp during storage and before planting. |
The broad, often mottled, mid-Green marbled purplish- Erythroniums fit in naturally with Trilliums, Galanthus, Hepatica, Helleborus, Hosta, Pulmonaria, Cyclamen coum and Cyclamen hederifolium. |
Grow under deciduous trees/shrubs, in a rock garden, or naturalize in thin grass. Ideally they like a soil which will dry out in Summer although many will do very well in a normal shady bed or border. Must receive adequate moisture during early spring when the foliage is making growth. Appreciates additional dressings of fallen leaves when the plant is in woodland gardens. |
Erythroniums do best when planted under trees and shrubs - to provide partial shade during the hottest part of the day, in as near to a woodland setting as possible. Plant bulbs 5 inches (12.5 cms) deep in good, rich soil; in the autumn in soil that does not dry out. If you want to plant them in pots use a John Innes compost rather than a peat based compost. They will be fine in this and should only be repotted when it is absolutely necessary. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Erythronium |
Sulphur-Yellow with brown central rings
|
April, May, June Forms a large Clump. |
12 x 4 Chalk, |
Bronze-mottled, glossy, deep green |
Plant in pots, woodland or under shrubs in bed. Use as indoor plant in Green-house or sunny window of cool room inside house. Inside Alpine House, or outside in Alpine Trough, or Window-box. |
Bulbs must be kept slightly damp during storage and before planting. A good variety to start off with. Received an 'Award of Merit' in 1959. Ideal compost is equal parts loam, peat, leaf mould and sand for pots. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Erythronium |
Bright Yellow
|
April, May, June Forms a large clump. |
12 x 4 Chalk, |
Wavy-margined, pale to mid-green. |
Plant in pots, woodland or under shrubs in bed. Use as indoor plant in Green-house or sunny window of cool room inside house. |
Plant inside Alpine House, or outside in Alpine Trough, or Window-box. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Dark Brown, Maroon and Black
|
October, November, December 6 petal, star-shaped flowers |
16-20 x 16 ( 40-50 x 40) Well-drained Sand, Scree Suitable for coastal conditions in stony or sandy soil. |
Light green leaves overlap each other being up to 12 inches long, with the uppermost surrounding the flowers.
|
The corms should be planted 3-4 inches (7.5-10 cms) deep and 6-8 inches (15-20 cms) apart in pots in a frost-free greenhouse during the winter and then the pots can be sunk into a south-facing rock garden during the summer in bold clumps. |
It grows in dunes and sandy places in South Africa. Flowers may last only one day, but the plant will continue to produce flowers for several weeks from October to early December. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Freesia "For outdoor culture, any light rich sandy soil will suffice, and the bulbs should be planted 2 inch (5 cms) deep and 2 inches apart in August and September. Do not move plants while growing as plants resent being disturbed. "Pot not more than 5 top-sized corms into a 5 inch (12.5 cm) pot from August and onwards, using John Innes compost or 4 parts sand, 3 parts leaf-mould with 0.5 ounces medium bone-meal mixed in the compost. The pots should then be plunged in a sunny spot in the garden, or frame, and remain there until there is the first possibility of frosts. During this time the corms must develop a good length of leaf. Where there is no garden a peat-filled box set up by a sunny window will do as a plunging ground. In such case it is important to see that the peat is kept sufficiently moist and that the excessive heat through the window does not scorch the potting compost. The window should be kept open in hot weather and at all convenient times. |
The following details come from Cactus Art:- "A flower is the the complex sexual reproductive structure of Angiosperms, typically consisting of an axis bearing perianth parts, androecium (male) and gynoecium (female). Bisexual flower show four distinctive parts arranged in rings inside each other which are technically modified leaves: Sepal, petal, stamen & pistil. This flower is referred to as complete (with all four parts) and perfect (with "male" stamens and "female" pistil). The ovary ripens into a fruit and the ovules inside develop into seeds. Incomplete flowers are lacking one or more of the four main parts. Imperfect (unisexual) flowers contain a pistil or stamens, but not both. The colourful parts of a flower and its scent attract pollinators and guide them to the nectary, usually at the base of the flower tube.
Androecium (male Parts or stamens) Gynoecium (female Parts or carpels or pistil) It is made up of the stigma, style, and ovary. Each pistil is constructed of one to many rolled leaflike structures. Stigma This is the part of the pistil which receives the pollen grains and on which they germinate. Style This is the long stalk that the stigma sits on top of. Ovary The part of the plant that contains the ovules. Ovule The part of the ovary that becomes the seeds. Petal The colorful, often bright part of the flower (corolla). Sepal The parts that look like little green leaves that cover the outside of a flower bud (calix). (Undifferentiated "Perianth segment" that are not clearly differentiated into sepals and petals, take the names of tepals.)"
The following details come from Nectary Genomics:- "NECTAR. Many flowering plants attract potential pollinators by offering a reward of floral nectar. The primary solutes found in most nectars are varying ratios of sucrose, glucose and fructose, which can range from as little a 8% (w/w) in some species to as high as 80% in others. This abundance of simple sugars has resulted in the general perception that nectar consists of little more than sugar-water; however, numerous studies indicate that it is actually a complex mixture of components. Additional compounds found in a variety of nectars include other sugars, all 20 standard amino acids, phenolics, alkaloids, flavonoids, terpenes, vitamins, organic acids, oils, free fatty acids, metal ions and proteins. NECTARIES. An organ known as the floral nectary is responsible for producing the complex mixture of compounds found in nectar. Nectaries can occur in different areas of flowers, and often take on diverse forms in different species, even to the point of being used for taxonomic purposes. Nectaries undergo remarkable morphological and metabolic changes during the course of floral development. For example, it is known that pre-secretory nectaries in a number of species accumulate large amounts of starch, which is followed by a rapid degradation of amyloplast granules just prior to anthesis and nectar secretion. These sugars presumably serve as a source of nectar carbohydrate. WHY STUDY NECTAR? Nearly one-third of all worldwide crops are dependent on animals to achieve efficient pollination. In addition, U.S. pollinator-dependent crops have been estimated to have an annual value of up to $15 billion. Many crop species are largely self-incompatible (not self-fertile) and almost entirely on animal pollinators to achieve full fecundity; poor pollinator visitation has been reported to reduce yields of certain species by up to 50%." |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Freesia alba |
White
|
March, 6 petal, funnel-shaped flowers in a cluster. Very strongly scented. |
8-17 x 4 Sand, or potting compost, |
Light Green sword-like leaves
|
Bring pot indoors when nightime temperature drops below 9 degrees Centigrade. Excellent house plants and cut flowers. |
Native to South Africa. Main attraction with these bulbs is the sweet fragrance that fills the room. If outside, mulch in autumn, remove mulch in summer. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Freesia andersoniae |
Cream to Purple with yellow.
|
April, May. 6 petal, funnel-shaped flowers in a cluster. Very fragrant. |
8 x 4 Sand, Gravel, or potting compost, |
Dark Green
|
Bring pot indoors during autumn and winter Excellent house plants and cut flowers, also in rock garden next to house wall. |
Native to southern coastal areas of South Africa. Plant against South-facing House Wall in Southern England |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Pale yellow with bright yellow-orange markings.
|
April, May. |
12 x 24 Sand, Gravel, or potting compost, |
Erect, spiral dark green fan, 10 inches (25 cms) long.
|
Bring pot indoors during autumn and winter. Excellent house plants and cut flowers, also in rock garden next to house wall. |
Native to eastern Cape Province of South Africa. Plant against South-facing House Wall in Southern England |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Fragrant Ivory White with mauve reverse and yellow markings.
|
April, May. 6 petal, funnel-shaped flowers in a cluster. |
6-12 x 6 Sand, Gravel, or potting compost, |
Dark Green
|
Bring pot indoors during autumn and winter. Excellent house plants and cut flowers, also in rock garden next to house wall. |
Native to South Africa. In colder areas, lift corms after foliage dies, store overwinter, and replant in the spring. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Fragrant Ivory-White
|
April, May. 6 petal, funnel-shaped flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Bring pot indoors during autumn and winter. Excellent house plants and cut flowers, also in rock garden next to house wall. |
Introduced in 1957 and recei-ved an 'Award of Merit' in 1962. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Fragrant Ivory-White.
|
April, May. 6 petal, funnel-shaped flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Dusky Pink on a
|
April, May. 6 petal, funnel-shaped double-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Dark Red on a pale
|
April, May. 6 petal, funnel-shaped single-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lemon Yellow.
|
April, May. 6 petal, funnel-shaped single-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape |
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Yellow.
|
April, May. 6 petal, funnel-shaped double-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
White.
|
April, May. 6 petal, funnel-shaped double-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Red.
|
April, May. 6 petal, funnel-shaped single-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Creamy-White.
|
April, May. 6 petal, funnel-shaped double-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Buttercup Yellow.
|
April, May. 6 petal, funnel-shaped single-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Purple-Red.
|
April, May. 6 petal, funnel-shaped semi-double-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Yellow.
|
April, May. 6 petal, funnel-shaped single-flowered flowers in a cluster. |
10 x 20 Sand, Gravel, or potting compost, |
Dark Green foliage held in fan shape
|
Excellent house plants and cut flowers, also in rock garden next to house wall. |
Bring pot indoors during autumn and winter. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Blue Bird' - tender |
Pale Blue and Purple
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm
|
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Castor' - tender |
Violet Purple
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm
|
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia flexuosa - tender |
Pinkish Mauve
|
June, July Clump. |
24 x 24 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm |
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Giant' - tender |
Ivory and Purple
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm |
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Hogarth' - tender |
Cream and Purple
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm |
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Holland's Gloire' |
Yellow
|
July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm
|
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Mabel' - tender |
Pink with Red Blush
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm
|
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia maculata - tender |
Yellow with Purplish-
|
May, June Clump. |
18 x 12 |
4 erect, narrow, sword-shaped, dark green leaves per corm
|
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Marquette' - tender |
Yellow and Purple
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm |
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Rose Emperor' |
Pink with
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm
|
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Titia' - tender |
Magenta
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm
|
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Venus' - tender |
Dark Red
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm |
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Vulcan' - tender |
Pink and Purple
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm
|
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ixia 'Yellow Emperor' |
Yellow with
|
June, July Clump. |
16 x 12 |
3-5 erect, narrow, sword-shaped, dark green leaves per corm
|
Grow in greenhouse, cool conserv-atory, patio pot, raised rock garden by south facing wall, window-box. Ground cover |
In very mild areas, plant out in sandy soil with good drainage and 1 inch (2.5 cms) deep coarse bark mulch, in March and then lift in late summer when the foliage has died down. Then, corms should be allowed to become dry. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia aloides - |
Green, Crimson and
|
March, April, May 3 petal, |
10 x 12 Sand or potting compost, |
2 broad-to-lanceolate leaves which are dark green with purple markings
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia aloides |
Yellow
|
March, April, May 3 petal, |
10 x 12 Sand or potting compost, |
2 broad-to-lanceolate leaves which are dark green with purple markings
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia aloides |
Red, Yellow, Green
|
March, April, May 3 petal, |
8-12 x 12 Sand or potting compost, |
2 broad-to-lanceolate leaves which are dark green with purple markings
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia aloides |
Bright Orange edged
|
March, April, May 3 petal, |
12-16 x 12 (30-40 x 30) Sand or potting compost, |
Mid-Green foliage and flower stems with brown markings
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia aloides |
Greenish-White
|
March, April, May 3 petal, |
4-8 x 12 Sand or potting compost, |
2 broad-to-lanceolate leaves which are dark green with purple markings
|
Edging in frost-free gardens. Very robust |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia bulbifera |
Coral-Red edged with Green or Purple
|
March, April, May 3 petal, |
6-15 x 12 Sand or potting compost, |
2 broad-to-lanceolate leaves which are dark green with purple spots
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
These are the galleries that will provide the plants to be added to their own Extra Index Pages
The following Extra Index of Bulbs is created in the
Having transferred the Extra Index row entry to the relevant Extra Index row for the same type of plant in a gallery below; then
|
Index of Bulbs from Further details on bulbs from the Infill Galleries:-
---------
|
Bulbs and Corms with
Index of Bulbs from
Website Structure Explanation and
There are other pages on Plants which bloom in each month of the year in this website :-
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia contaminata |
White with Maroon tips and stripes
|
April, May 3 petal, |
6 x 12 Sand or potting compost, |
Grass-like in appearance and plain Green
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia elegans var. suaveolens |
Blue shading to Rose
|
March, April, May 3 petal, |
7-9 x 12 (17.5-22.5 x 30) Sand or potting compost, |
Mid-Green
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Fransie' |
Pink shading to Yellow with Maroon tips
|
March, April, May 3 petal, |
12 x 12 Sand or potting compost, |
Mid-Green foliage with mid-Green stems spotted Purple
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia glaucina var. pallida |
Cream with a Yellow or Pale Green Hue
|
March, April, May 3 petal, |
8 x 12 Sand or potting compost, |
Dark Green foliage slightly mottled Purple with pale Green flower stems
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia juncifolia |
White tinged Red
|
March, April, May 3 petal, |
6 x 12 Sand or potting compost, |
Mid-Green foliage with mid-Green stems
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Namakwa' |
Orange fading to Yellow, with Pink tips
|
March, April, May 3 petal, |
12 x 12 Sand or potting compost, |
Mid-Green foliage with Orange flower stems
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia namaquensis |
Blue shading to Magenta, White internally
|
March, April, May 3 petal, |
6-8 x 12 Sand or potting compost, |
Mid-Green foliage
|
Edging in frost-free gardens. |
Spreads rapidly by means of long stoloniferous roots. Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Nova' |
Bluish-Green
|
March, April, May 3 petal, |
8 x 12 Sand or potting compost, |
Mid-Green foliage with Purple flower stems
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia orthopetala |
White
|
March, April, May 3 petal, |
10 x 12 Sand or potting compost, |
Mid-Green grassy foliage with Purple flower stems
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia pustulata |
Cream or Pale Yellow, to Pink or Blue
|
March 3 petal, |
12 x 12 Sand or potting compost, |
Mid-Green foliage with Purple flower stems
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Robyn' |
Red
|
March, April, May 3 petal, |
12 x 12 Sand or potting compost, |
Mid-Green foliage with Purple flower stems
|
Edging in frost-free gardens. |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. They can be grown as bed edging in only Southern England, Isle of Wight and Channel Islands within the UK. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Rolina' |
Creamy-Yellow
|
March, April, May 3 petal, |
12 x 12 |
Mid-Green with Purple flower stems
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Romaud' |
Buttercup-Yellow with Creamy-White tips
|
March, April, May 3 petal, |
12 x 12 |
Mid-Green with Purple flower stems
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Romelia' |
Light Yellow
|
March, April, May 3 petal, |
12 x 12 |
Mid-Green with Purple flower stems
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Ronina' |
Yellow
|
March, April, May 3 petal, |
12 x 12 |
Mid-Green with Purple flower stems
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Rosabeth' |
Red outer petals, inside is Yellow
|
March, April, May 3 petal, |
12 x 12 |
Mid-Green with Purple spots
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia rosea |
Blue through to Pink
|
March, April, May 3 petal, |
10 x 12 |
Mid-Green
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia 'Rupert' |
Lilac-Purple
|
March, April, May 3 petal, |
12 x 12 |
Mid-Green
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia splendida |
Blue shaded Lilac
|
March, April, May 3 petal, |
10 x 12 |
Light Green
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia unifolia |
White with Blue shading
|
May 3 petal, |
10-12 x 12 (20-30 x 30) |
Light Green
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia viridiflora |
Blue-Green to Turquoise
|
March, April, May 3 petal, |
8 x 12 |
Mid-Green
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Lachenalia zeyheri |
White,
|
March, April, May 3 petal, |
4-8 x 12 |
Mid-Green
|
Edging in frost-free gardens. Houseplant in Patio Pot within a sunny but unheated room. Patio Pot or |
Use either John Innes compost or a mixture of 2 parts sandy loam, 0.5 part leaf-mould and 0.5 part decayed manure, with 1 part coarse sand in pots or hanging baskets. Will not tolerate frost so grow in Greenhouse or as houseplant in a sunny but unheated room. Bed edging in only Southern England. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Leucocoryne 'Andes' |
Mauve with Purple
|
May, June 6 petal, umbel-shaped flower in an umbellate. Sweetly scented. |
10-14 x 4 (25-35 x 10) Well-drained Sand or potting compost, |
Grass-like green foliage varies in length from 6-12 inches. Often they are just maturing or even have died down by going yellow by the time the first flowers are seen. |
A small genus of only 12 species from the winter rainfall regions of South America. These make excellent pot plants in a frost-free greenhouse or unheated room in the house, and |
Plant in the sloping ground next to a South-facing wall in the Channel Islands or in pots in cold frame or greenhouse for the remainder of the UK. The bulbs will not tolerate frost. This plant is resistant to deer! |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Leucocoryne 'Caravelle' |
Mauve with Plum
|
April 6 petal, umbel-shaped flower in an umbellate. Sweetly scented. |
12-16 x 4 (30-40 x 10) Well-drained Sand or potting compost, |
Grass-like green foliage varies in length from 6-12 inches. Often they are just maturing or even have died down by going yellow by the time the first flowers are seen. |
A small genus of only 12 species from the winter rainfall regions of South America. These make excellent pot plants in a frost-free greenhouse or unheated room in the house, and |
Plant in the sloping ground next to a South-facing wall in the Channel Islands or in pots in cold frame or greenhouse for the remainder of the UK. The bulbs will not tolerate frost. This plant is resistant to deer! |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Massonia echinata |
White fading to Pink
|
February Tubular flower |
2 x 10 Well-drained sand or potting mix, |
2 wide green leaves about 5 inches long, which lie flat on the ground.
|
Makes an attractive and unusual late winter flowering pot. Full Sun in a Conservatory in the UK, where temperatures do not fall below 45 degrees Fahrenheit (7 degess Centigrade) in pot or hanging basket |
In well-drained soil (sand) in rock garden within Channel Islands where temperature exceeds 7C, otherwise grow in mixture of 2 parts topsoil, 3 parts peat moss and 7 parts sharp builder's sand in wide pots. Place shards of broken clay pots in the bottom to ensure good drainage. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Mela-sphaerula ramosa Zones 8-10 of Hardiness Zone Map developed by the United States Department of Agriculture (USDA) |
Pale Yellow
|
April, May, June Tall dainty Gypsophila-like stems are covered with 6 narrow finely pointed petals in small starry flowers within a spray. |
12 x 3-6 Well-drained sand or potting mix, Part Shade in a Conservatory in the UK, where temperatures do not fall below 45 degrees Fahrenheit. Moist in growth, dry in dormancy |
Long, narrow, light green leaves up to !0 inches (25 cms) in length. |
Suits pot cultivation in UK Conservatory. Lasts very well as cut flower. The bulbs will not tolerate frost. Moisture is necessary at the time of planting in late July-September, but keep barely moist until the foliage is observed. Then, additional amounts of water should be given, but never allow the bulbs to sit in cold, wet soil. Should be a complete resting period in the summer with dry conditions. |
Plant 1 inch (2.5 cms) deep and 3-6 inches (7.5-15 cms) apart in the ground next to a South-facing wall in the Channel Islands or 5 bulbs per 10 inch (25 cms) pot and 1 inch (2.5 cms) deep. Soil - In well-drained frost-free soil (sand) perhaps in Channel Islands, otherwise grow in mixture of 2 parts topsoil, 3 parts peat moss and 7 parts sharp builder's sand in wide pots. Place shards of broken clay pots in the bottom to ensure good drainage. Grows in sheltered damp places among rocks in southern Africa. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Magenta-Pink with
|
September |
8 x 4 |
Light green clover-like foliage, often twisting and closing at night or on very hot days. The foliage is not present during the late autumn and winter, when the plant is dormant. |
An outstanding selection with magenta-pink funnel-shaped flowers with yellow throats held above light green clover-like foliage in early autumn. Frost tender, so one for the greenhouse. |
This is good for hanging baskets. Plant 1 inch (2.5 cms) deep and 3-4 inches (7.5-10 cms) apart. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Reddish-Purple with
|
September, October, |
0.5-2 x 4 (1.25-5 x 10) |
Light green clover-like foliage, often twisting and closing at night or on very hot days. The foliage is not present during the late autumn and winter, when the plant is dormant. |
This is good for hanging baskets. Plant 1 inch (2.5 cms) deep and 3-4 inches (7.5-10 cms) apart. |
Oxalis is an enormous family of plants from all over the globe. These in this Gallery are a selection of winter-growing varieties. All are easy to grow and very rewarding with very long flowering times. There are approximately 1919 species. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Yellow
|
May, June, July |
4 x 4 |
Small tufts of light green clover-like leaves appear in spring and then die down for several months, before re-appearing in early autumn at the same time as the bright yellow funnel-shaped flowers. Foliage is absent in the winter. Mat-forming habit. Deep mulch after autumn foliage has died down to prevent the bulb being frozen. |
This is good for hanging baskets. Plant 1 inch (2.5 cms) deep and 3-4 inches (7.5-10 cms) apart. |
Frost hardy, this will withstand temperatures down to -5c. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Telos Rare Bulbs in USA have other Oxalis varieties for sale from |
Pink with a Yellow centre
|
May, June, July |
10 x 10 |
Light green clover-like foliage with a silver gloss, often twisting and closing at night or on very hot days. The foliage is not present during the late autumn and winter, when the plant is dormant. |
This is a variable winter-growing oxalis from South Africa which produces delicate flowers in a range of pinks and apricots which last for ages. Do not feed to keep the leaves contained. This is suitable between paving, massed at the front of a low border or in a wall and rock garden, also suitable for window-boxes. Plant 1 inch (2.5 cms) deep and 3-4 inches (7.5-10 cms) apart. |
Oxalis are wonderful "collector's items" -- you know you have been bitten by the bug when, upon seeing their dazzling jewel-like flowers and different leaf forms, you experience an irrepressible urge to possess more! The South African species are largely winter-growers, brightening the dreary months with their exuberant flowers, then go dormant in summer. They are best appreciated as container plants, and need sun to open their flowers. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
White
|
October, November |
5 x 2 |
Light Green, erect, 0.25 inches wide and 4-5 inches high, foliage |
Polyxena is a small family of very dwarf bulbs suited to pot culture in a frost-free situation. Can start to flower in the autumn soon after potting. Very uncommon and well worth growing. This is suitable for hanging baskets in the summer and in coldframes for the rest of the year. Plant 1 inch (2.5 cms) deep and 2-3 inches (5-7.5 cms) apart. Can be grown outside in the Channel Islands in sandy soil. Moisture is needed in early spring, with little or none needed after the foliage dies back in late autumn. |
Small, white flowers are held between the leaves with flower fragrance much like that of a hyacinth. The native habitat in Cape Province of South Africa is open, sparse grassland near the coast. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Deep Lilac
|
October, November |
2-3 x 12 (5-7.5 x 30) |
Light Green, erect, 1 inch wide and 4-6 inches in length, foliage |
This bulb has clusters of starry-like deep lilac flowers produced at the base of the strappy green foliage. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Sparaxis grandiflora acutiloba - tender
Sparaxis is derived from the Greek "sparasso" ("to tear"), which refers to the lacerated spathes that surround the flowers |
Golden-Yellow
|
April, May |
4-10 x 12 (10-25 x 30) |
Flat, stiff and rather tough dark Green leaves 8 inches long are held in a fan shape at the base of the flowering spike. |
This is suitable for hanging baskets in the summer and in coldframes for the rest of the year where they can be protected from the frost below 25 degrees Fahrenheit. Plants will withstand a few degrees of frost, but not prolonged cold temperatures. Plant 2 inches (5 cms) deep and 3-4 inches (7.5-10 cms) apart. Can be grown outside in the Channel Islands in sandy soil in bold groups of 25 or more in one place. Moisture is needed in early spring, with none needed after the foliage dies back in late autumn, so that the corms ripen. Great cut flowers, as they are long-lasting. |
Sparaxis, native to South Africa, has been in cultivation for over 200 years, due to its ease and free flowering form. As part of the Iris family, brightly coloured flowers are borne above the strappy foliage. Colours range from hot oranges, yellows and pinks to reds and dark purple. Well worth a pot display in fertile gritty loam under frost free conditions. The plants prefer to be on the dry side in the summer as in their native habitats of South Africa, where they receive their rainfall in the winter. |
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Deep Violet with
|
April, May |
6-12 x 12 (15-30 x 30) |
Flat, stiff and rather tough dark Green leaves 8 inches long are held in a fan shape at the base of the flowering spike. |
This is suitable for hanging baskets in the summer and in coldframes for the rest of the year where they can be protected from the frost below 25 degrees Fahrenheit. Plants will withstand a few degrees of frost, but not prolonged cold temperatures. Plant 2 inches (5 cms) deep and 3-4 inches (7.5-10 cms) apart. Can be grown outside in the Channel Islands in sandy soil in bold groups of 25 or more in one place. Moisture is needed in early spring, with none needed after the foliage dies back in late autumn, so that the corms ripen. Great cut flowers, as they are long-lasting. |
Sparaxis, native to South Africa, has been in cultivation for over 200 years, due to its ease and free flowering form. As part of the Iris family, brightly coloured flowers are borne above the strappy foliage. Colours range from hot oranges, yellows and pinks to reds and dark purple. Well worth a pot display in fertile gritty loam under frost free conditions. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Yellow and Cream with Purple flush
|
April, May, June |
6-12 x 12 (15-30 x 30) |
Flat, stiff and rather tough dark Green leaves 8 inches long are held in a fan shape at the base of the flowering spike. |
This is suitable for hanging baskets in the summer and in coldframes for the rest of the year where they can be protected from the frost below 25 degrees Fahrenheit. Plants will withstand a few degrees of frost, but not prolonged cold temperatures. Plant 2 inches (5 cms) deep and 3-4 inches (7.5-10 cms) apart. Can be grown outside in the Channel Islands in sandy soil in bold groups of 25 or more in one place. Moisture is needed in early spring, with none needed after the foliage dies back in late autumn, so that the corms ripen. Great cut flowers, as they are long-lasting. |
Sparaxis, native to South Africa, has been in cultivation for over 200 years, due to its ease and free flowering form. As part of the Iris family, brightly coloured flowers are borne above the strappy foliage. Colours range from hot oranges, yellows and pinks to reds and dark purple. Well worth a pot display in fertile gritty loam under frost free conditions. |
Functional combinations in the border from the International Flower Bulb Centre in Holland:- "Here is a list of the perennials shown by research to be the best plants to accompany various flower bulbs. The flower bulbs were tested over a period of years in several perennial borders that had been established for at least three years. In combination with hyacinths:
In combination with tulips:
In combination with narcissi:
For narcissi, the choice was difficult to make. The list contains only some of the perennials that are very suitable for combining with narcissi. In other words, narcissi can easily compete with perennials. In combination with specialty bulbs:
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Red, Orange, and Yellow to White with Red and Gold or Black throat
|
May, June, July |
12 x 16 |
Flat, stiff and rather tough dark Green leaves 10 inches long and 0.33 inches wide are held in a fan shape at the base of the flowering spike. |
This corm has Six-petalled flowers, which are produced on wiry stems in early to mid-summer in a wide range of colours from red, orange and yellow to white. In addition some have a very striking red and gold or black throat. The foliage is narrow and strap-like, up to 25cm long. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Pale Red
|
May, June |
9 x 16 |
The stiff, pointed, sword-shaped leaves are held in a basal fan and are shorter than the flower spike. |
Tritonia is a small genus of corms from South Africa. Bright flowers are arranged along wiry stems, borne above the grassy foliage. These make a lovely cut flower. The varieties listed in this Gallery are from winter growing regions and so are best cultivated in pots in a frost free situation. Pale Red flowers are erect and bowl-shaped, 1.5 inches in diameter |
This is suitable for hanging baskets in the summer and in coldframes for the rest of the year where they can be protected from the frost below 25 degrees Fahrenheit. Plants will withstand a few degrees of frost, but not prolonged cold temperatures. Plant 2 inches (5 cms) deep and 4-6 inches (10-15 cms) apart. Can be grown outside in the Channel Islands in sandy soil in bold groups of 25 or more in one place in a rock garden. Moisture is needed in early spring, with none needed after the foliage dies back in late summer, so that the corms ripen. In the wild of Cape Province in South Africa, they are found growing in grassy areas where there is considerable moisture during the growing season, followed by a drier period. Great cut flowers, as they are long-lasting. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
White
|
May, June |
9 x 16 |
The stiff, pointed, sword-shaped leaves are held in a basal fan and are shorter than the flower spike. |
This corm has "pure white bowl-shaped flowers. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Pink
|
May, June |
10 x 16 |
The stiff, pointed, sword-shaped leaves are held in a basal fan and are shorter than the flower spike. |
This corm has very pretty pink flowers. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Pale Red
|
May, June |
10 x 16 |
The stiff, pointed, sword-shaped leaves are held in a basal fan and are shorter than the flower spike. |
This corm has pale red flowers. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Orange
|
May, June |
10 x 16 |
The stiff, pointed, sword-shaped leaves are held in a basal fan and are shorter than the flower spike. |
This corm has hot orange flowers. |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The flower stalk is mottled with Purple and is about 18-20 inches in height. Pale Rose and flecked at the tip with Green |
December, January,
|
18 x 30 |
About 10 basal leaves are produced, each up to 18 inches long and 4 inches wide, with undulating margins, forming a rosette. Sometimes flecked with pale green, contrasting well with the shiny deep green. |
This bulb is one for a sunny windowsill or warm greenhouse but well worth growing. A rosette of long fleshy leaves are produced, from the middle of which a single tall flower spike grows. Up to 50 pink, tubular flowers can be borne, the insides are often spotted yellow. Need a minimum of 5 degrees Centigrade (41degrees Fahrenheit). |
Veltheimia bracteata is a native of western areas of the Cape Province of South Africa. This is suitable as a house pot plant. Make sure the containers are large enough so that they can grow for awhile without being repotted. Plant 1 inch (2.5 cms) deep and 6-10 inches (15-25 cms) apart. |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Site design and content copyright ©June 2007. Page structure amended November 2012. DISCLAIMER: Links to external sites are provided as a courtesy to visitors. Ivydene Horticultural Services are not responsible for the content and/or quality of external web sites linked from this site. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
UKButterflies Larval Foodplants website page lists the larval foodplants used by British butterflies. The name of each foodplant links to a Google search. An indication of whether the foodplant is a primary or secondary food source is also given. Please note that the Butterfly you see for only a short time has grown up on plants as an egg, caterpillar and chrysalis for up to 11 months, before becoming a butterfly. If the plants that they live on during that time are removed, or sprayed with herbicide, then you will not see the butterfly. |
||||
Plants used by the Butterflies follow the Plants used by the Egg, Caterpillar and Chrysalis as stated in |
||||
Plant Name |
Butterfly Name |
Egg/ Caterpillar/ Chrysalis/ Butterfly |
Plant Usage |
Plant Usage Months |
Egg, |
1 egg under leaf. |
10 days in May-June |
||
Egg, |
Eggs laid in batches encircling the branch of the food plant. |
Hatches after 18-22 days in April. |
||
Egg, |
Groups of eggs on upper side of leaf. |
- |
||
Egg, |
1 egg at base of plant. |
Late August-April |
||
Egg, |
Groups of eggs on upper side of leaf. |
- |
||
Egg, |
1 egg laid on underside of leaflets or bracts. |
7 days in June. |
||
Egg, |
1 egg laid on underside of leaflets or bracts. |
7 days in June. |
||
Egg, |
1 egg laid under the leaf or on top of the flower. |
7 days in August. |
||
Egg, |
1 egg on underside of a flower bud on its stalk. |
7 days. |
||
Egg, |
1 egg on underside of a flower bud on its stalk. |
7 days. |
||
Egg, |
1 egg under leaf. |
10 days in May-June. |
||
Egg, |
1 egg on leaf. |
2 weeks |
||
Cabbages - Large White eats all cruciferous plants, such as cabbages, mustard, turnips, radishes, cresses, nasturtiums, wild mignonette and dyer's weed |
Egg,
|
40-100 eggs on both surfaces of leaf. |
May-June and August-Early September. 4.5-17 days. |
|
Egg, |
1 egg on underside of leaf. |
May-June and August. 7 days. |
||
Cabbages:- |
Egg, |
1 egg on underside of leaf. |
July or August; hatches in 3 days. |
|
Cabbages:- |
Egg, |
1 egg laid in the tight buds and flowers. |
May-June 7 days. |
|
Cherry with |
Egg, |
Eggs laid in batches encircling the branch of the food plant. |
Hatches after 18-22 days in April. |
|
Egg, |
Groups of eggs on upper side of leaf. |
- |
||
Egg, |
1 egg on leaf. |
10 days in May-June. |
||
Egg, |
1 egg on leaf. |
6 days in May-June. |
||
Egg, |
1 egg under leaf. |
|
||
|
(Common CowWheat, Field CowWheat) |
Egg, |
Eggs laid in batches on the under side of the leaves. |
Hatches after 16 days in June. |
|
Currants |
Egg, |
Groups of eggs on upper side of leaf. |
|
|
Egg, |
Eggs laid in batches on the under side of the leaves. |
Hatches after 20 days in July. |
||
Dog Violet with |
Egg, |
1 egg on oak or pine tree trunk |
15 days in July. |
|
Dog Violet with |
Egg, |
1 egg on leaf or stem. |
Hatches after 15 days in May-June. |
|
Dog Violet with |
Egg, |
1 egg on leaf or stem. |
Hatches after 10 days in May-June. |
|
Egg, |
1 egg on underside of a flower bud on its stalk. |
7 days. |
||
Egg, |
Eggs laid in batches encircling the branch of the food plant. |
Hatches after 18-22 days in April. |
||
False Brome is a grass (Wood Brome, Wood False-brome and Slender False-brome) |
Egg, |
1 egg under leaf. |
... |
|
Egg, |
Eggs laid in batches on the under side of the leaves. |
Hatches after 20 days in July. |
||
Egg, |
1 egg laid on underside of leaflets or bracts. |
7 days in June. |
||
Egg, |
1 egg on leaf or stem. |
Hatches after 10 days in May-June. |
||
Egg, |
1 egg on underside of a flower bud on its stalk. |
7 days. |
||
Egg, |
1 egg laid under the leaf or on top of the flower. |
7 days in August. |
||
Egg, |
1 egg on leaf. 5 or 6 eggs may be deposited by separate females on one leaf. |
14 days in July-August. |
||
Egg, |
1 egg on underside of a flower bud on its stalk. |
7 days. |
||
Egg, |
1 egg laid in the tight buds and flowers. |
May-June 7 days. |
||
Egg, |
Eggs laid in batches on the under side of the leaves. |
Hatches after 20 days in July. |
||
Egg, |
Groups of eggs on upper side of leaf. |
|
||
Egg, |
1 egg under leaf. |
1 then |
||
Egg, |
1 egg on underside of a flower bud on its stalk. |
7 days. |
||
Egg, |
1 egg at base of plant. |
Late August-April. |
||
Egg, |
1 egg on leaf. |
10 days in May-June. |
||
Egg, |
1 egg on leaf. |
2 weeks |
||
Egg, |
1 egg on leaf. |
6 days in May-June. |
||
Egg, |
1 egg on underside of leaf. |
May-June and August. 7 days. |
||
Egg, |
1 egg on leaf. 5 or 6 eggs may be deposited by separate females on one leaf. |
14 days in July-August. |
||
Narrow-leaved Plantain (Ribwort Plantain) |
Egg, |
Eggs laid in batches on the under side of the leaves. |
Hatches after 16 days in June. |
|
Narrow-leaved Plantain (Ribwort Plantain) |
Egg, |
Eggs laid in batches on the under side of the leaves. |
Hatches after 16 days in June. |
|
Nasturtium from Gardens |
Egg, |
1 egg on underside of leaf. |
May-June and August. 7 days. |
|
Egg, |
1 egg on tree trunk |
15 days in July. |
||
Mountain pansy, |
Egg, Chrysalis |
1 egg laid under the leaf or on top of the flower. |
7 days in August. 3 weeks in September |
|
Egg, |
1 egg on tree trunk. |
15 days in July. |
||
Egg, |
Eggs laid in batches on the under side of the leaves. |
Hatches after 20 days in July. |
||
Egg, |
Eggs laid in batches encircling the branch of the food plant. |
Hatches after 18-22 days in April. |
||
Egg, |
Groups of eggs on upper side of leaf. |
- |
||
Egg, |
1 egg under leaf. |
|
||
Egg, |
1 egg laid under the leaf or on top of the flower. |
7 days in August. |
||
Egg, |
Eggs laid in batches encircling the branch of the food plant. |
Hatches after 18-22 days in April. |
||
Egg, |
Eggs laid in batches on the under side of the leaves. |
Hatches after 16 days in June. |
||
Egg, |
1 egg on underside of a flower bud on its stalk. |
7 days. |
||
Egg, |
1 egg on underside of a flower bud on its stalk. |
7 days. |
||
Egg, |
Groups of eggs on upper side of leaf. |
|
||
Egg, |
1 egg under leaf. |
|
||
Egg, |
1 egg on leaf. |
2 weeks |
||
Trefoils 1, 2, 3 |
Egg, |
1 egg on leaf. |
6 days in May-June. |
|
Egg, |
Groups of eggs on upper side of leaf. |
- |
||
Egg, |
1 egg laid on underside of leaflets or bracts. |
7 days in June. |
||
Violets:- |
Egg, |
1 egg on underside of leaf or on stalk. |
July-August for 17 days. |
|
Violets:- |
Egg, |
1 egg on stem or stalk near plant base. |
July to hatch in 8 months in March. |
|
Egg, |
1 egg on leaf. |
2 weeks. |
||
Egg, |
Eggs laid in batches encircling the branch of the food plant. |
Hatches after 18-22 days in April. |
||
Egg, |
1 egg on leaf. 5 or 6 eggs may be deposited by separate females on one leaf. |
14 days in July-August. |
||
Willow |
Egg, |
Eggs laid in batches encircling the branch of the food plant. |
Hatches after 18-22 days in April. |
|
Egg, |
Eggs laid in batches on the under side of the leaves. |
Hatches after 20 days in July. |
||
Plants used by the Butterflies |
||||
Plant Name |
Butterfly Name |
Egg/ Caterpillar/ Chrysalis/ Butterfly |
Plant Usage |
Plant Usage Months |
Asters |
Butterfly |
Eats nectar. |
|
|
Runner and Broad Beans in fields and gardens |
Butterfly |
Eats nectar |
April-June or July-September. |
|
Aubretia in gardens |
Butterfly |
Eats nectar |
May-June or August till killed by frost and damp in September-November |
|
Butterfly |
Eats sap exuding from trunk. |
April-Mid June and Mid July-Early September for second generation. |
||
Butterfly |
Eats nectar. |
20 days. |
||
Butterfly |
Eats nectar |
May-June |
||
Holly Blue |
Butterfly |
Eats nectar |
April-Mid June and Mid July-Early September for second generation. |
|
Butterfly |
Eats nectar. |
July-October. |
||
Buddleias |
Butterfly |
Eats nectar. |
July-October. |
|
Wood White |
Butterfly |
Eats nectar |
May-June. |
|
Cabbage and cabbages in fields |
Butterfly |
Eats nectar |
April-June or July-September. |
|
Butterfly |
Eats nectar |
July-October |
||
Adonis Blue |
Butterfly |
Eats nectar. |
1 Month during Mid-May to Mid-June or during August-September |
|
Pale Clouded Yellow |
Butterfly |
Eats nectar |
May-June or August till killed by frost and damp in September-November |
|
Cow-wheat |
Butterfly |
Eats nectar |
June-July |
|
Butterfly |
Eats nectar |
May-June |
||
Butterfly |
Eats nectar |
April-Mid June and Mid July-Early September for second generation. |
||
Butterfly |
Eats nectar. |
3 weeks between May and September |
||
Germander Speedwell (Veronica chamaedrys - Birdseye Speedwell) |
Butterfly |
Eats nectar |
June-July |
|
Butterfly |
Eats nectar. |
July-October. |
||
Butterfly |
Eats nectar |
30 days in May-June. |
||
Butterfly |
Eats nectar |
May-September |
||
Butterfly |
Eats nectar. |
May-June for 18 days. |
||
Butterfly |
Eats nectar. |
July-October |
||
Butterfly |
Eats nectar. |
1 Month. |
||
Butterfly |
Eats nectar. |
July-October. |
||
Painted Lady |
Butterfly |
Eats nectar |
July-October. |
|
Marigolds in gardens |
Butterfly |
Eats nectar |
May-June or August till killed by frost and damp in September-November |
|
Butterfly |
Eats nectar. |
1 Month during Mid-May to Mid-June or during August-September. |
||
Michaelmas Daisies |
Butterfly |
Eats nectar. |
July-October |
|
Butterfly |
Eats nectar |
April-June or July-September. |
||
Narrow-leaved Plantain (Ribwort Plantain) |
Butterfly |
Eats nectar |
June-July |
|
Nasturtiums in gardens |
Butterfly |
Eats nectar |
April-June or July-September |
|
Butterfly |
Eats sap exuding from trunk. |
April-Mid June and Mid July-Early September for second generation. |
||
Butterfly |
Eats nectar |
June. |
||
Butterfly |
Eats nectar |
May-June. |
||
Butterfly |
Eats nectar |
July-October. |
||
Butterfly |
Eats nectar |
July-May |
||
Butterfly |
Eats nectar |
7 weeks in July-August. |
||
Comma |
Butterfly |
Eats nectar. |
July-October. |
|
Butterfly |
Eats nectar. |
3 weeks between May and September |
||
Trefoils 1, 2, 3 |
Butterfly |
Eats nectar. |
1 Month during Mid-May to Mid-June or during August-September |
|
Butterfly |
Eats nectar. |
20 days in August. |
||
Butterfly |
Eats nectar |
June.
|
||
Butterfly |
Eats nectar |
June-July |
||
Apple/Pear/Cherry/Plum Fruit Tree Blossom in Spring |
Butterfly |
Eats Nectar |
April-May |
|
Rotten Fruit |
Butterfly |
Drinks juice |
July-September |
|
Tree sap and damaged ripe fruit, which are high in sugar |
Butterfly |
Hibernates inside hollow trees or outhouses until March. Eats sap or fruit juice until April. |
10 months in June-April |
|
Wild Flowers |
Large Skipper |
Butterfly |
Eats Nectar |
June-August |
Links to the other Butterflies:- Black Hairstreak |
Topic - Wildlife on Plant Photo Gallery. Some UK native butterflies eat material from UK Native Wildflowers and live on them as eggs, caterpillars (Large Skipper eats False Brome grass - Brachypodium sylvaticum - for 11 months from July to May as a Caterpillar before becoming a Chrysalis within 3 weeks in May) chrysalis or butterflies ALL YEAR ROUND. |
Wild Flower Family Page (the families within "The Pocket Guide to Wild Flowers" by David McClintock & R.S.R. Fitter, Published in 1956 They are not in Common Name alphabetical order and neither are the common names of the plants detailed within each family. The information in the above book is back-referenced to the respective page in "Flora of the British Isles" by A.R. Clapham of University of Sheffield, |
||
When you look at the life history graphs of each of the 68 butterflies of Britain, you will see that they use plants throughout all 12 months - the information of what plant is used by the egg, caterpillar, chrysalis or butterfly is also given in the above first column.
THE LIFE AND DEATH OF A FLAILED CORNISH HEDGE - This details that life and death from July 1972 to 2019, with the following result:- End note, June 2008. I hear spring vetch has been officially recorded somewhere in West Cornwall and confirmed as a presence in the county, so perhaps I can be permitted to have seen it pre-1972 in the survey mile. I wonder where they found it? It's gone from hedges where it used to be, along with other scarcities and so-called scarcities that used to flourish in so many hedges unrecorded, before the flail arrived. I have given careful thought to including mention of some of the plants and butterflies. So little seems to be known of the species resident in Cornish hedges pre-flail that I realise some references may invite scepticism. I am a sceptic myself, so sympathise with the reaction; but I have concluded that, with a view to re-establishing vulnerable species, it needs to be known that they can with the right management safely and perpetually thrive in ordinary Cornish hedges. In future this knowledge could solve the increasingly difficult question of sufficient and suitable sites for sustainable wild flower and butterfly conservation - as long as it is a future in which the hedge-flail does not figure.
CHECK-LIST OF TYPES OF CORNISH HEDGE FLORA by Sarah Carter of Cornish Hedges Library:-
Titles of papers available on www.cornishhedges.co.uk:-
THE GUILD OF CORNISH HEDGERS is the non-profit-making organisation founded in 2002 to support the concern among traditional hedgers about poor standards of workmanship in Cornish hedging today. The Guild has raised public awareness of Cornwall's unique heritage of hedges and promoted free access to the Cornish Hedges Library, the only existing source of full and reliable written knowledge on Cornish hedges." |
||||
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Recommended Plants for Wildlife in different situations
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
From the Ivydene Gardens Box to Crowberry Wild Flower Families Gallery: |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The Bumblebee Pages website is divided into five major areas:
FORCED INDOOR BULBS in Window Box Gardens. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Theme |
Plants |
Comments |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Thyme |
Thymus praecox, wild thyme Thymus pulegioides Thymus leucotrichus Thymus citriodorus |
Thymes make a very fragrant, easy to care for windowbox, and an excellent choice for windy sites. The flower colour will be pinky/purple, and you can eat the leaves if your air is not too polluted. Try to get one variegated thyme to add a little colour when there are no flowers. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Herb |
Sage, mint, chives, thyme, rosemary |
Get the plants from the herb section of the supermarket, so you can eat the leaves. Do not include basil as it need greater fertility than the others. Pot the rosemary up separately if it grows too large. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Mints |
Mentha longifolia, horse mint Mentha spicata, spear mint Mentha pulgium, pennyroyal Mentha piperita, peppermint Mentha suaveolens, apple mint |
Mints are fairly fast growers, so you could start this box with seed. They are thugs, though, and will very soon be fighting for space. So you will either have to thin and cut back or else you will end up with one species - the strongest. The very best mint tea I ever had was in Marrakesh. A glass full of fresh mint was placed in front of me, and boiling water was poured into it. Then I was given a cube of sugar to hold between my teeth while I sipped the tea. Plant this box and you can have mint tea for months. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Heather |
Too many to list See Heather Shrub gallery |
For year-round colour try to plant varieties that flower at different times of year. Heather requires acid soils, so fertilise with an ericaceous fertilser, and plant in ericaceous compost. Cut back after flowering and remove the cuttings. It is best to buy plants as heather is slow growing. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Blue |
Ajuga reptans, bugle Endymion non-scriptus, bluebell Myosotis spp., forget-me-not Pentaglottis sempervirens, alkanet |
This will give you flowers from March till July. The bluebells should be bought as bulbs, as seed will take a few years to flower. The others can be started from seed. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Yellow |
Anthyllis vulneraria, kidney vetch Geum urbanum, wood avens Lathryus pratensis, meadow vetchling Linaria vulgaris, toadflax Lotus corniculatus, birdsfoot trefoil Primula vulgaris, primrose Ranunculus acris, meadow buttercup Ranunculus ficaria, lesser celandine |
These will give you flowers from May to October, and if you include the primrose, from February. Try to include a vetch as they can climb or trail so occupy the space that other plants can't. All can be grown from seed. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
White |
Trifolium repens, white clover Bellis perennis, daisy Digitalis purpurea alba, white foxglove Alyssum maritimum Redsea odorata, mignonette |
All can be grown from seed. The clover and daisy will have to be cut back as they will take over. The clover roots add nitrogen to the soil. The mignonette flower doesn't look very special, but the fragrance is wonderful, and the alyssum smells of honey. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Pink |
Lychnis flos-cucli, ragged robin Scabiosa columbaria, small scabious Symphytum officinale, comfrey |
The comfrey will try to take over. Its leaves make an excellent fertiliser, and are very good on the compost heap, though windowbox gardeners rarely have one. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Fragrant |
Lonicera spp., honeysuckle Alyssum maritimum Redsea odorata, mignonette Lathyrus odoratus, sweet pea |
The sweet pea will need twine or something to climb up, so is suitable if you have sliding windows or window that open inwards. You will be rewarded by a fragrant curtain every time you open your window. |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Spring bulbs and late wildflowers |
Galanthus nivalis, snowdrop Narcissus pseudonarcissus, narcissius Crocus purpureus, crocus Cyclamen spp. |
The idea of this box is to maximize your space. The bulbs (cyclamen has a corm) will flower and do their stuff early in the year. After flowering cut the heads off as you don't want them making seed, but leave the leaves as they fatten up the bulbs to store energy for next year. The foliage of the wildflowers will hide the bulb leaves to some extent. Then the wildflowers take over and flower till autumn |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Aster spp., Michaelmas daisy Linaria vulgaris, toadflax Lonicera spp., honeysuckle Succisa pratensis, devil's bit scabious Mentha pulgium, pennyroyal |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Bee Garden in Europe or North America |
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Plants for moths (including larval food plants and adult nectar sources) from Gardens for Wildlife - Practical advice on how to attract wildlife to your garden by Martin Walters as an Aura Garden Guide. Published in 2007 - ISBN 978 1905765041:- |
Marjoram - Origanum officinale |
"On average, 2 gardeners a year die in the UK as a result of poisonous plants. Those discussed in this blog illustrate a range of concerns that should be foremost in the designer’s mind." from Pages on poisonous plants in this website:- |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Wildlife-friendly Show Gardens
Many of our gardens at Natural Surroundings demonstrate what you can do at home to encourage wildlife in your garden:-
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Ivydene Gardens Water Fern to Yew Wild Flower Families Gallery: |
Only Wildflowers detailed in the following Wildflower Colour Pages |
|
|||||||||||||||
Jan |
Feb |
Mar |
Apr |
May |
Jun |
Jul |
Aug |
Sep |
Oct |
Nov |
Dec |
||||||
1 |
Blue |
||||||||||||||||
1 |
|||||||||||||||||
1 |
Cream |
||||||||||||||||
1 |
|||||||||||||||||
1 |
|||||||||||||||||
1 |
|||||||||||||||||
1 |
|||||||||||||||||
1 |
|||||||||||||||||
1 |
|||||||||||||||||
1 |
|||||||||||||||||
1 |
White A-D |
||||||||||||||||
1 Yellow |
|||||||||||||||||
1 |
|||||||||||||||||
1 |
|||||||||||||||||
1 |
|||||||||||||||||
1 |
Flowering plants of |
||||||||||||||||
1 |
Flowering plants of |
||||||||||||||||
Copied from Botanical Index Gallery Pages |
Appended to Botanical Name is |
Number of Botanical Plants in each Page or Gallery / comparison pages section |
||
A, B, C, D, E, Links to 1000's of Indexed Plants in the galleries below are in addition to the ones above:- Bee pollinated plants per flower colour per month in Bee-Pollinated Alpines, Aquatic, Annual, Beddi-ng, Biennial and Bulb with Climber of 3 sector system are in
Botanical Wildflowers in |
Plant Type:- Gr = Grass |
Flower Colour:- followed by Annual, Bulb, Climber, |
Inv = Invasive; so pot the plant instead |
A, 391 Galleries/Comparison Pages:- Total 5851 plus plants in the above Galleries / Comparison pages section |
How comparison thumbnails with their text boxes will be changed to from
Flower Colour and Foliage Colour if not green Violet-blue, ACIDIC SAND, LOAM. Above contents of this text box require amend-ing Soil. |
||||